Immune Response to Vaccines in Dialysis Patients: Covid-19 and Beyond
Authors:
PD Dr. med. Olivier Giannini
Viceprimario Medicina Interna
Ente Ospedaliero Cantonale (EOC)
Ospedale Regionale di Mendrisio, Beata Vergine
Via Turconi 23
6850 Mendrisio
E-Mail: olivier.giannini@eoc.ch
Dr. sc. Luca Piccoli
Associate Director
Humabs BioMed SA, a subsidiary of Vir Biotechnology
Via dei Gaggini 3
6500 Bellinzona
E-Mail: lpiccoli@vir.bio
Sie sind bereits registriert?
Loggen Sie sich mit Ihrem Universimed-Benutzerkonto ein:
Sie sind noch nicht registriert?
Registrieren Sie sich jetzt kostenlos auf universimed.com und erhalten Sie Zugang zu allen Artikeln, bewerten Sie Inhalte und speichern Sie interessante Beiträge in Ihrem persönlichen Bereich
zum späteren Lesen. Ihre Registrierung ist für alle Unversimed-Portale gültig. (inkl. allgemeineplus.at & med-Diplom.at)
Patients on dialysis have often shown an impaired response to different vaccines, whose mechanism has not been fully elucidated. However, Covid-19 vaccines have provided a unique opportunity to deeply analyze the immune response in this population. Most recent data indicate that seroconversion rates may not reflect the real neutralizing activity of vaccine-induced antibodies. Therefore, continuous immune monitoring with proper serological tests is crucial to adopt the best prevention and therapeutic strategy.
Keypoints
-
Patients with end-stage kidney disease (ESKD), who are highly predisposed to life-threatening infections, show a reduced response to vaccines.
-
Despite a variable seroprotection rate, influenza vaccination results in clinical effectiveness in this population.
-
Vaccination against HBV is not effective in a sizable fraction of dialysis patients and their survival can be easily estimated using seroconversion and seroprotection parameters.
-
Covid-19 mRNA vaccines induce high rates of seroconversion, which are, unfortunately, accompanied by low levels of rapidly declining neutralizing antibodies against SARS-CoV-2 variants, especially in dialysis patients that have never been exposed to the virus.
Patients with end-stage kidney disease (ESKD) are highly predisposed to infections, which are the major causes of morbidity and the second cause of mortality in this vulnerable population. Many studies showed an overall reduced response to different vaccines, including those against Influenza virus and HBV. It has long been established that uraemia is associated with an acquired immunodeficiency state induced by dysregulation of the immune system. Although not completely elucidated, complex and multifactorial causes, including dysfunction in neutrophils, macrophages, B cells, T cells and in complement activation are involved in the immune dysregulation observed in ESKD. Paradoxically, concomitant signs of activation of immune cells have been described in dialysis patients, which are further accentuated rather than corrected by renal replacement treatments.1 Nevertheless, a delayed and impaired antibody response and rapid waning of immunity to different vaccines, including those against HBV, suggests the importance of continuous immune monitoring and specific booster protocols in these patients.
Infections by SARS-CoV-2 have posed a new threat for ESKD patients, who were found to have a greater risk of severe Covid-19 course. The rapid development of Covid-19 vaccines has provided, on one side, an important strategy to prevent SARS-CoV-2 infection in these patients, and, on the other side, a unique opportunity to deeply analyse the immune response to vaccines in this frail population. Most recent data about the immune response induced by vaccination against Influenza virus, HBV and SARS-CoV-2 will be critically analysed and discussed in this review.
Influenza Vaccination
Even if there is still limited data on the prevalence and mortality rate of ESKD patients following Influenza infection, both the US guidelines set by the Centers for Disease Control and Prevention (CDC) and those of the European Centre for Disease Prevention and Control similarly recommend annual flu vaccination as a reasonable policy.2 The current vaccination rates in the dialysis population are about 70%, based on the 2013–2014 flu season data and have improved markedly over the last 10 years.2,3 Most studies on vaccine efficacy consider a fourfold increase in hemagglutinin antibody response protective from infection.2 Indeed, seroconversion was observed in a percentage of patients that varies from 20% in older studies to 80% in more recent ones. In the majority of these studies, immune protection was assessed approximately one month after the administration of the vaccine, while other studies assessed vaccine efficacy 6 month after vaccination.3
Although the mechanism of suboptimal antibody response to the flu vaccination has not been elucidated yet, a 2-year analysis of the US Medicare data found that vaccinated patients on dialysis had a substantially lower risk of any-cause hospital admission and death compared to unvaccinated patients.4 A large US-Taiwanese cohort study of patients on hemodialysis (HD) showed that those receiving the flu vaccination have a reduced risk of influenza-associated pneumonia, ICU hospitalization as well as a lower mortality rate, in particular for the elderly.5 In another study, after excluding patients with less than 1 year on dialysis and those whose vaccination status was unknown, the relative risk of all-cause mortality was reduced of approximately 30% in those receiving the flu vaccination and nearly 40% in those receiving both influenza and pneumococcal vaccines.6 This finding might indicate clinical effectiveness in vaccinating this population despite a variable seroprotection rate.
Hepatitis B Virus (HBV) Vaccination
HBV vaccination in patients with ESKD is widely recommended by regulatory agencies, including the CDC, to curtail the risk of outbreaks of HBV infection in dialysis facilities.3 In the early ’80s, first reports showed that only 50% of recipients of patients on HD were considered as vaccine responders.7 In a more recent meta-analysis of 61 studies, non-responders were found to be 30% of patients cumulatively analysed.8 The differences in the HBV vaccine effectiveness observed in the different studies are most likely due to different vaccine formulations, doses and vaccination programs.
Different risk factors, including older age, diabetes mellitus and immunosuppressive treatments, have been identified for a reduced vaccination response in both the general population and dialysis patients.8 This meta-analysis performed on a large number of dialysis patients (6628) showed that HBV-vaccine responders tend to be younger than non-responders, with a lower prevalence of diabetes mellitus and better nutritional and inflammatory markers, more adequate dialysis markers, higher intact parathormone (iPTH) levels and are less likely to carry the HLA-DR3 susceptibility gene. However, achieving seroconversion and seroprotection (defined as a anti-HBs titre ≥10IU/L and ≥100IU/L, respectively) after active HBV vaccination was shown to be associated with a significantly reduced all-cause and cardiovascular-related mortality.9 This observation suggests that patient survival can be easily estimated using these simple and readily available parameters independently of other well-known key parameters, including age, gender, the presence of diabetes and markers of malnutrition and inflammation.
The mechanism of HBV vaccine unresponsiveness in CKD patients has not been completely elucidated so far. Different cellular defects in both B cell and T cell compartments have been identified, including low absolute cell counts and reduced activation of specific cell subsets, and have been partially attributed to the metabolic unbalance caused by uraemia and hyperphosphatemia. In particular, it has been shown that HBV vaccination elicited a poor plasmablast response and impaired activation of CXCR3+CCR6–CCR7– subset of circulating T follicular helper cells that might explain the impaired seroconversion observed in non-responding CKD patients.10
Covid-19 Vaccine
From the early beginning of the Covid-19 pandemic, it was clear that infection by SARS-CoV-2 would have posed a new threat for CKD patients, in particular for those with ESKD on dialysis. Indeed, these patients were rapidly found to have a greater risk of severe Covid-19 courses, with a mortality rate ranging from 10% to 30%.11 In addition, the rapid increase of cases of more infective SARS-CoV-2 variants, in particular Delta and Omicron, provided a new potential challenge for dialysis patients.
The rapidly developed Covid-19 vaccines have been fundamental to prevent SARS-CoV-2 infections, especially in frail individuals. Among these, patients on dialysis were quickly prioritized in international vaccination programs. However, dialysis patients, as in general all immunosuppressed subjects, showed a lower antibody response to vaccines, in particular after the first and second dose, compared to healthy individuals. Serological studies in HD patients showed a delayed and reduced response to vaccines and also a rapid decline of anti-SARS-CoV-2 antibodies titres.12,13 This finding is reminiscence of the low response observed after flu and HBV vaccinations, as described above. However, most serological studies have described the impaired immune response to Covid-19 vaccines by defining antibody titres to SARS-CoV-2 Spike (S) protein, without measuring the neutralizing activity of vaccine-induced antibodies, which is a more important correlate of protection.14
In a recent study,15 we performed a deep characterization of the serum antibody response in 143 dialysis patients (130 HD and 13 on peritoneal dialysis [PD]), who were enrolled from the four dialysis units of the Ente Ospedaliero Cantonale (EOC) in Southern Switzerland and received two doses of a Covid-19 mRNA vaccine (either BNT162b2 from Pfizer-BioNTech or mRNA-1273 from Moderna) between January and May 2021. Most patients (83%) had never been exposed to SARS-CoV-2 before the vaccination, whereas the remaining 17% reported a previous infection with SARS-CoV-2, with severe Covid-19 symptoms (75%) that required hospitalization (54%) or admission to an intensive care unit (21%). Anti-SARS-CoV-2 S antibodies were measured in 94.4% of dialysis patients after the second vaccine dose but 35% of them had still low or undetectable levels of antibodies, compared to 100% of 48 healthcare workers, who were included as healthy controls (HC).
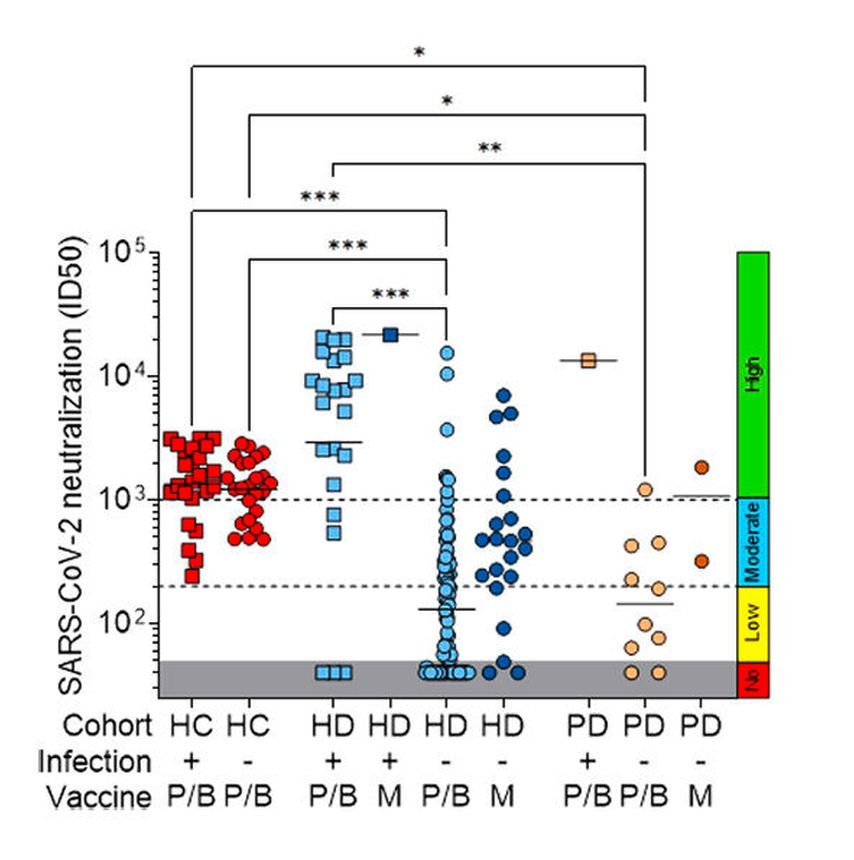
Fig. 1: Neutralizing antibody titres (ID50: 50% of inhibitory dilution) against wild-type SARS-CoV-2 pseudoviruses (reported from Bassi et al.)15
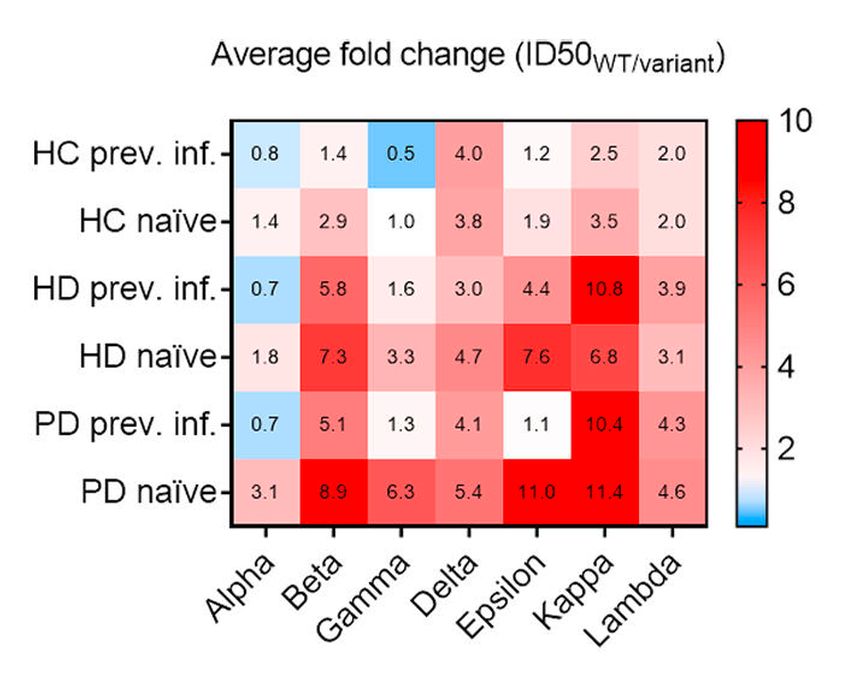
Fig. 2: Fold change analysis of loss of neutralizing titers against different variants (reported from Bassi et al.)15
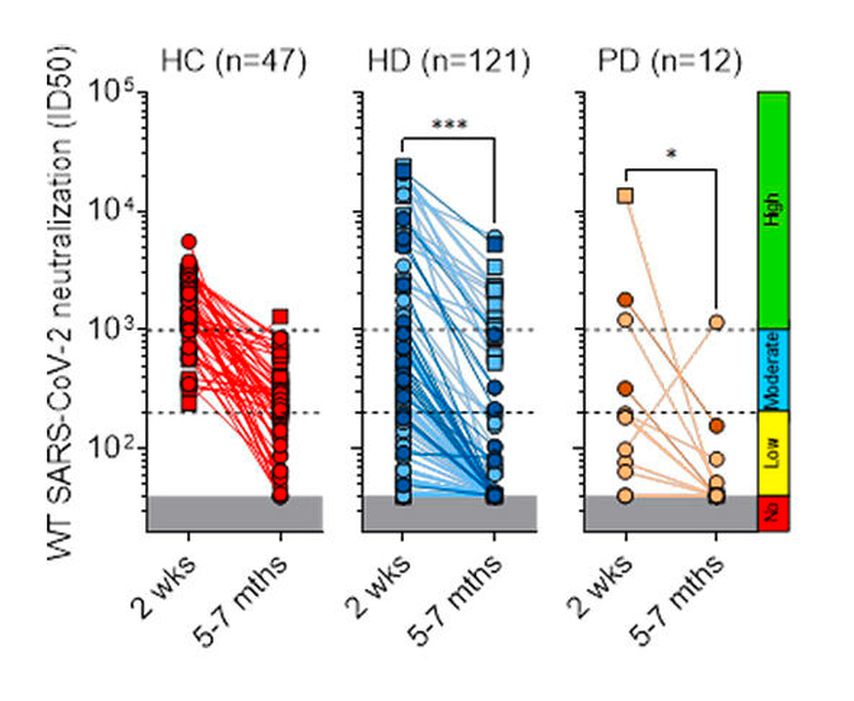
When we analysed the neutralizing activity of serum antibodies induced by vaccination we found that 49% of patients had low-level or undetectable neutralizing antibodies against the vaccine-matched SARS-CoV-2, with a significant reduction in neutralizing titres against other variants (Fig. 1 and 2). In particular, the fraction of dialysis patients who poorly responded to the vaccine further increased to 77% in the case of the Delta variant. Of note, the fraction of non-responding patients was found to be higher in SARS-CoV-2-naïve HD patients immunized with Pfizer-BioNTech (66%) than those immunized with Moderna (23%). In addition, follow-up data, collected up to 7 months after vaccination, showed a rapid decay of neutralizing antibodies to both wild-type SARS-CoV-2 and Delta variant (Fig. 3). Taken together, these data suggest that two doses of an mRNA vaccine may not be sufficient for dialysis patients to develop a protective antibody response against SARS-CoV-2 variants. These patients have, therefore, an increased risk of infection with possible severe outcomes over time from vaccination, as suggested by a recent study on a unit veteran cohort followed up to 6 months after vaccination.16 Nevertheless, during the follow-up, we could not identify any new case of SARS-CoV-2 infection in our cohort, an observation that may reflect the very low incidence recorded in Switzerland in the spring-summer 2021 season, as well as the adherence of the dialysis patients to the strict prevention rules.
Fig. 3: Side-by-side comparison of neutralizing titres measured 2 weeks and up to 7 months after the second Covid-19 vaccine dose (reported from Bassi et al.)15
Among different parameters analysed, including socio-demographic and clinical data, we identified that having never been exposed to SARS-CoV-2 was the major factor that increased the risk of a poor antibody response after vaccination, especially in HD patients immunized with Pfizer-BioNTech. This finding is consistent with another study showing that healthy naïve individuals develop lower titres of neutralizing antibodies to SARS-CoV-2 variants compared to previously infected individuals after two mRNA vaccine doses17 and suggests that vaccination may boost the neutralizing antibody response in dialysis patients after infection.18 Similarly to other studies, we also found other risk factors of poor neutralizing antibody response to mRNA vaccines, including age, heart failure and immunosuppression.
Although not fully elucidated, the mechanism underlying the poor response to Covid-19 vaccines in dialysis patients may be explained by a delayed and slow affinity maturation of SARS-CoV-2 S-specific B cells. Indeed, it was shown that specific B cells were enriched among pre-switch and naïve B cells in dialysis patients, whereas healthy controls showed higher fractions of plasmablasts and post-switch B cells.19 Consistent with these data, our analyses showed that healthy controls exhibited a higher antibody avidity that correlated with neutralizing activity, as compared to naïve dialysis patients, who instead showed a poorly neutralizing and low-avidity response.
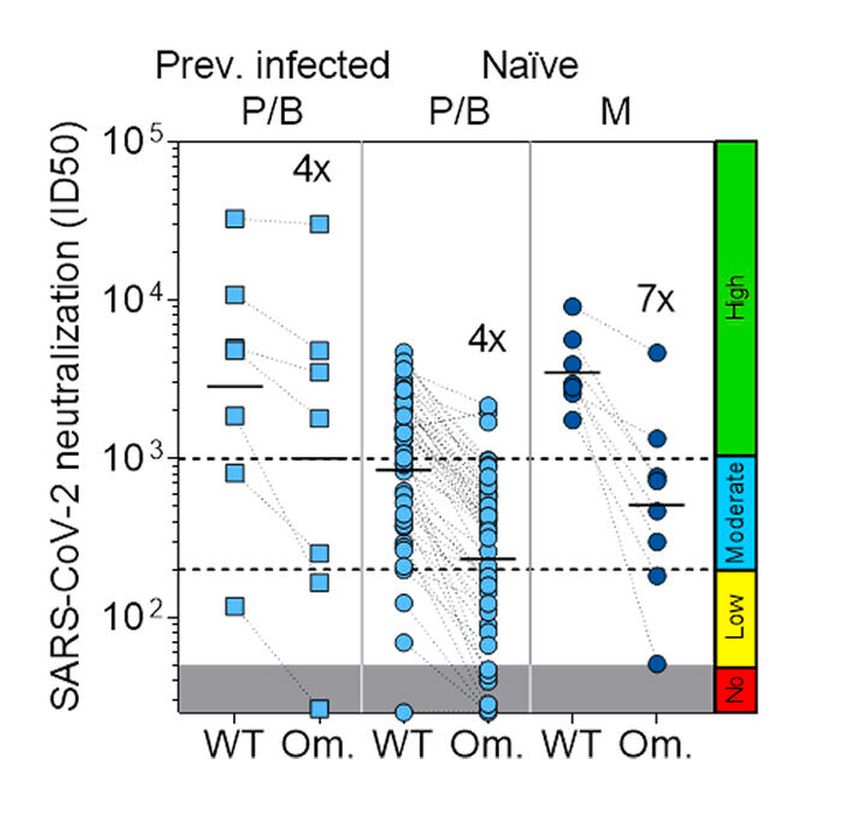
Collectively, these data indicate that the standard two-dose scheme of vaccination might provide insufficient protection to dialysis patients against SARS-CoV-2, especially in the case of a highly transmissible variant, such as Omicron, and support the need for additional boosts, in particular for non-responding patients. Efficacy of a third dose of BNt162B2 vaccine has been shown in previously unresponsive older adults as well as in dialysis patients.20,21 In a recent study, we also supported these findings by showing that more than 90% of dialysis patients produced neutralizing antibodies against wild-type and Omicron SARS-CoV-2 after receiving a third dose of BNt162B2 or mRNA1273 vaccine (Fig.4).22
Fig. 4: Side-by-side comparison of neutralizing titres against wild-type (WT) and Omicron (Om.) SARS-CoV-2 pseudoviruses. Average fold-loss on Omicron is shown (adapted from Cameroni et al.)22
Concluding Remarks and Outlook
In conclusion, vaccinations against Influenza virus, HBV and SARS-CoV-2 have demonstrated high safety and sufficient efficacy in reducing severe outcomes in dialysis patients. However, the immune response to vaccines of a sizable fraction of patients is defective compared to healthy individuals. It is, therefore, important, in order to adopt the best prophylactic and therapeutic strategy, that we continuously monitor the immune response of our patients on dialysis after vaccination by using proper serological tests that measure not only the serum antibody levels, but, when possible, also their neutralizing activity, either directly or indirectly through an avidity test. Moreover, additional studies are needed to better understand the immune dysfunction and indirect immunodepression of dialysis patients and more generally in patients with ESKD.
Literature:
1 Descamps-Latscha B: The immune system in end-stage renal disease. Curr Opin Nephrol Hypertens 1993; 2: 883-91 2 Bowman BT, Rosner MH: Influenza and the patient with end-stage renal disease. J Nephrol 2018; 31: 225-30 3 Kunisaki KM, Janoff EN: Influenza in immunosuppressed populations: a review of infection frequency, morbidity, mortality, and vaccine responses. Lancet Infect Dis 2009; 9: 493-504 4 Gilbertson DT et al.: Influenza vaccine delivery and effectiveness in end-stage renal disease. Kidney Int 2003; 63: 738-43 5 Wang IK et al.: Effectiveness of influenza vaccination in patients with end-stage renal disease receiving hemodialysis: a population-based study. PLoS One 2013; 8: e58317 6 Bond TC et al.: Mortality of dialysis patients according to influenza and pneumococcal vaccination status. Am J Kidney Dis 2012; 60: 959-65 7 Stevens CE et al.: Hepatitis B vaccine in patients receiving hemodialysis. Immunogenicity and efficacy. N Engl J Med 1984; 311: 496-501 8 Udomkarnjananun S et al.: Hepatitis B virus vaccine immune response and mortality in dialysis patients: a meta-analysis. J Nephrol 2020; 33: 343-54 9 Zitt E et al.: Response to active hepatitis B vaccination and mortality in incident dialysis patients. Vaccine 2017; 35: 814-20 10 da Silva EN et al.: A randomized trial of serological and cellular responses to hepatitis B vaccination in chronic kidney disease. PLoS One 2018; 13: e0204477 11 Jager KJ et al.: Results from the ERA-EDTA Registry indicate a high mortality due to COVID-19 in dialysis patients and kidney transplant recipients across Europe. Kidney Int 2020; 98: 1540-48 12 Rincon-Arevalo H et al.: Impaired humoral immunity to SARS-CoV-2 BNT162b2 vaccine in kidney transplant recipients and dialysis patients. Sci Immunol 2021; 6: eabj1031 13 Grupper A et al.: Humoral response to the Pfizer BNT162b2 vaccine in patients undergoing maintenance hemodialysis. Clin J Am Soc Nephrol 2021; 16: 1037-42 14 Khoury DS et al.: Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat Med 2021; 27: 1205-11 15 Bassi J et al.: Poor neutralization and rapid decay of antibodies to SARS-CoV-2 variants in vaccinated dialysis patients. PLoS One 2022; 17: e0263328 16 Cohn BA et al.: SARS-CoV-2 vaccine protection and deaths among US veterans during 2021. Science 2022; 375: 331-6 17 McCallum M et al.: SARS-CoV-2 immune evasion by the B.1.427/B.1.429 variant of concern. Science 2021; 373: 648-54 18 Chan L et al.: Antibody response to mRNA-1273 SARS-CoV-2 vaccine in hemodialysis patients with and without prior COVID-19. Clin J Am Soc Nephrol 2021; 16: 1258-60 19 Rincon-Arevalo H et al.: Impaired humoral immunity to SARS-CoV-2 BNT162b2 vaccine in kidney transplant recipients and dialysis patients. Sci Immunol 2021; 6: eabj1031 20 Romero-Olmedo AJ et al.: Induction of robust cellular and humoral immunity against SARS-CoV-2 after a third dose of BNT162b2 vaccine in previously unresponsive older adults. Nat Microbiol 2022; 7: 195-9 21 Espi M et al.: The ROMANOV study found impaired humoral and cellular immune responses to SARS-CoV-2 mRNA vaccine in virus-unexposed patients receiving maintenance hemodialysis. Kidney Int 2021; 100: 928-36 22 Cameroni E et al.: Broadly neutralizing antibodies overcome SARS-CoV-2 Omicron antigenic shift. Nature 2022; 602: 664-70
Das könnte Sie auch interessieren:
Nephrokalzinose, Mutationen, Nierensteine – und was dies mit dem Alter zu tun hat
Prof. Martin Konrad leitet die Pädiatrische Nephrologie an der Universitätsklinik für Kinder- und Jugendmedizin in Münster. An der Jahrestagung der Schweizerischen Gesellschaft für ...
Intensivierte Hämodialyse eröffnet neue Perspektiven
Eine Schwangerschaft galt bei Patientinnen mit terminaler Niereninsuffizienz jahrzehntelang als Ausnahme und war mit erheblichen Risiken für Mutter und Kind verbunden. Die intensivierte ...
Adipositas und ihre Folgen für die Niere
Die Adipositas ist zu einem der wichtigsten weltweiten Gesundheitsprobleme geworden. Diese hat direkte und indirekte Folgen für die Niere. Neben der Gefahr einer Glomerulopathie und der ...