©
Getty Images/iStockphoto
Promoting biological therapy in orthopaedic research
Leading Opinions
Autor:
Prof. Dr. phil. nat. Benjamin Gantenbein, PhD
E-Mail: benjamin.gantenbein@dbmr.unibe.ch<br> www.tom-lab.com
Autor:
Dr. Julien Guerrero, PhD
Department for BioMedical Research (DBMR)<br> Medical Faculty, University of Bern
30
Min. Lesezeit
05.03.2020
Weiterempfehlen
<p class="article-intro">As the population ages the demand for musculoskeletal replacement parts increases. Traditional surgery is often based on pure metallic and/or ceramics solutions. However, in many cases the biology of the human body is not respected. Tissue engineering could be in the lead for new solutions with better host integration, better range of motion, less inflammation, and lower reoperation rates.</p>
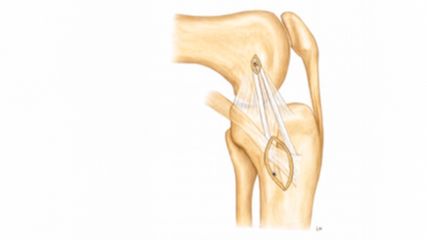
<p class="article-content"><div id="keypoints"> <h2>Keypoints</h2> <ul> <li>Due to an increasing elderly population the demand to understand the biology of musculoskeletal tissues has been increased.</li> <li>Hip implants represent the most successful story for orthopaedics with very low complication rates (around 2 % ).</li> <li>No good solution does yet exist for many joints affected by degenerative processes, such as artificial intervertebral disc implants, knee implants, ligament replacements.</li> <li>The current problems to be focused on in orthopaedic research: inflammation after surgery (complications), metabolic diseases affecting bone and joints (i. e. osteoporosis, ectopic ossification), long lasting pain that is often associated with a destabilizing spinal column.</li> <li>Progenitor cells isolated from adipose tissue or bone marrow might be a cell source for a biological cell therapy.</li> <li>Biomaterials are warranted that are instructive for specific cell phenotypes or may be combined with growth factors that maybe slowly released.</li> <li>The best cell source, however, may be the tissue-specific progenitor cells that reside in low numbers in all joint tissues.</li> </ul> </div> <h2>The unmet clinical needs to reduce low back pain</h2> <p>The intervertebral disc (IVD) has a very limited regenerative potential and disc degeneration is a major cause of chronic low back pain (LBP). This represents a leading cause of disability with significant economic and social burdens.<sup>1</sup> The total economic burden of low back pain (LBP) for the Swiss society was estimated between 1,6 % and 2,3 % of the gross domestic product (GDP).<sup>2</sup> The IVD consists of an inner nucleus pulposus tissue (NP) surrounded by the annulus fibrosus (AF) tissue, and hyaline articular cartilage located at the endplates between the IVD and the vertebral bodies. The gelatinous NP is an avascular tissue containing a highly organized extracellular matrix rich in proteoglycans and collagens with few dispersed cells. IVD degeneration implies a degradation of the extracellular matrix in the NP and the AF resulting in a reduced disc height. The exact mechanisms by which IVD degeneration is induced are still unknown. Some risk factors have been identified, they include ageing, genetic predisposition and stress factors. The degenerative changes of the IVD take place early in life and the rate of cellular turnover is much slower compared to other tissues.<br /> Current treatments aim to repair the degenerated disc by replacement of the injured tissue with a functional biological substitute or prosthesis. Conventional treatments of IVD degeneration are limited since conservative or surgical therapies do not restore the IVD tissue properties. Since the IVD possesses a very limited self-healing capacity, regenerative medicine by injection of cells may represent a promising therapy for treatment of disc degeneration. As such, IVD repair strategies require an appropriate cell source that is able to regenerate the damaged NP tissue, such as progenitor cells. Cell-based therapies by injection of IVD cells, chondrocytes, or progenitor cells have gained significant attention and progressed to clinical trials for treatment of spinal disorders.</p> <h2>The knee is a very complex joint and prone to problems</h2> <p>Rupture of the anterior cruciate ligament (ACL) is a widespread knee injury. The annual incidence in the US is estimated to lie between 100 000 to 200 000 cases.<sup>3</sup> ACL tears mostly occur as a result of rotational forces alongside varus/valgus stresses acting on the knee. Furthermore, about 80 % of all ACL incidences are so called «non-contact» injuries that happen due to unfortunate knee positioning and a strong unopposed quadriceps contraction. It is generally well accepted that ACL ruptures have a very poor self-healing capacity. Here, further research is required for a better understanding why ACL have such a poor wound-healing capacity. Non-successful healing of the ACL has the consequence of arthritis and non-physiological mechanical loading in the knee joint. Here, novel solutions are required that would accelerate the wound healing of the ACL and would prevent scar formation. Collagen patches wrapped around the ACL in combination with a dynamic fixation system could be in the lead for better wound healing. Here, investigations were done how these concepts may be applied in a clinically relevant ex-vivo set-up, where tendons or ligaments can be kept alive under physiological mechanical loading.</p> <h2>The current focus of the TOM research group</h2> <p>The Tissue Engineering for Orthopaedics and Mechanobiology group (short TOM group) of the Department for Orthopedics & Traumatology, Insel University Hospital, and the Department for Bio-Medical Research (DBMR), both University of Bern, conducts translational research in the intersection of tissue engineering, biology, and applied clinical research.<br /> The group’s primary aim is to understand the cellular response to biomechanical stimuli and how cellular communities are affected in situ using 3D tissue and organ culture models (Fig. 1). Our research can be divided into two main foci: On the one hand the group investigates causes of low back pain due to intervertebral disc degeneration and on the other hand the group focuses on the human knee and the shoulder with the aim to identify cell-based solutions for the non-healing or delayed rupture of the ACL and the rotator cuff of the shoulder. The common focus of the TOM group is to advance in-vitro organ culture models, which match closely the human situation and where regenerative therapy strategies, such as novel biomaterials and cells, can be tested in a most authentic in-vitro set-up (Fig. 1).<br /> Currently, the group investigates a particular progenitor-cell population that has been identified only very recently. These progenitors are positive for Tie2 marker (angiopoietin receptor-1). Our team showed that these cells are able to form colony-forming units (CFUs) and also can be differentiated into different lineages of the musculoskeletal apparatus, i. e., adipo-, osteo-, and chondrogenic lineages. This H2020-funded project called «iPSpine» aims to develop further the technology around induced progenitor cells (iPS cells) and to generate intervertebral disc cells de novo from various cell sources (see below).</p> <p><img src="/custom/img/files/files_datafiles_data_Zeitungen_2020_Leading Opinions_Ortho_2001_Weblinks_lo_ortho_2001_s11_fig1_gantenbein.jpg" alt="" width="550" height="479" /></p> <h2>Novel smart biomimetic materials are warranted – silk and hydrogels</h2> <p>In the past, the research team has been investigating actively smart biomaterials, which are biomimetic and should lead the way with new therapies. One of the oldest biomaterials with a very low inflammation response of the host is silk. Silk was applied in orthopaedics for many different applications, ranging from bone regeneration to tendon repair. Figure 2 visualizes the cyto- compatibility growing human primary cells isolated from the bone marrow or from the IVD tissue and how the cells interact with the silk biomaterial. Previous research in the TOM group investigated into new growth-factor enriched silk, which has been produced from genetically transduced silk worms (Bombyx mori), which embed the growth factor of interest (in this case growth and differentiation factor 6 = GDF6) directly into the silk. The new «advanced» biomaterial has then been tested in-vitro with disc cells and mesenchymal stem cells but also in an established 3D bovine organ culture model and the complex loading bioreactor together with a FDA-approved fibrin hydrogel. Therefore, a healthy control, an injured IVD (2 mm biopsy punch) and the repaired IVD were tested and histology was performed to visualize the injury and integration of the novel silk and fibrin hydrogel. These results were recently reported in the Journal of Orthopaedic Research and in the Journal of Functionalized Biomaterials.<sup>4, 5</sup> In a collaborative research project funded by the Swiss National Science Foundation (SNSF) and the German Research Funds (Deutsche Forschungsgemeinschaft = DFG) with Dr. Michael Wöltje, Technische Universität Dresden, Germany, it is planned to adapt the silk material towards the repair of the outer structure, i. e., the annulus fibrosus. Here, silk as a very traditional biomaterial will be used again but revamped with novel textile and fiber-reinforcement technologies for the application of annulus fibrosus repair.</p> <p><img src="/custom/img/files/files_datafiles_data_Zeitungen_2020_Leading Opinions_Ortho_2001_Weblinks_lo_ortho_2001_s12_fig2_gantenbein.jpg" alt="" width="550" height="574" /></p> <h2>iPSpine project</h2> <p>The consortium leading the iPSpine project was formed to initiate a European-led research effort to identify a future advanced therapeutic strategy that results in a radical new treatment of LBP induced by IVD degeneration. With their multidisciplinary expertise in the development of advanced therapies and their translation from bench to bedside, the aim of the iPSpine team is to investigate and develop a new advanced therapy medicinal product (ATMP) for the future, based on a novel developmental biology-based therapeutic strategy employing induced pluripotent stem cells (iPSC) and smart biomaterials. The iPSpine consortium will develop and demonstrate proof of concept with the aid of novel and extended knowledge, tools and technology platforms.<br /> The iPSpine impact: iPSpine seeks to offer novel technologies and ATMPs for the advanced therapy research and development community. IVD degeneration will be the showcase, offering improved quality of life for millions of patients with IVD degeneration-induced LBP, through long-lasting reduction of LBP, reduced LBP-related premature retirement, and improved socio-economic contribution.</p> <h2>A niche project</h2> <p>As degeneration of the NP is a major cause of IVD degeneration, research towards NP cells and NP progenitor cells is drawing increased attention. However, caused by difficulties associated with their harvest and culture, there are few reports investigating the culture methods of NP cells (NPCs) and the effects on the stemness of these cells.<br /> In this context, the ability to culture cells and to investigate the behaviour of NPCs in their microenvironment while maintaining their gene expression profile is essential in experiments using these cells. Therefore, understanding the cell morphology and gene expression profile of NPCs is critical for the development of new therapeutic strategies regarding IVD degeneration.<br /> It is well documented in the literature that the gene expression profile of human NPCs is easily lost with conventional two-dimensional (2D) monolayer culture, which does not replicate the three-dimensional (3D) natural environment. It is described as hydrophilic aggrecan-rich/ glycosaminoglycan-rich gelatinous extracellular matrix (ECM) with unique avascular, hypoxic, nutrient-deficient, high osmotic pressure characteristics in which NPCs are naturally supported. For this reason, to study them in a proper way, a novel culture method is required which allows for the number of NPCs to be expanded while maintaining their expression profile. In our project, we aimed to establish and to study novel, efficient primary culture and subsequent expansion culture methods for human NPCs.</p> <h2>Tie2 cells</h2> <p>The IVD has limited regenerative potential and disc degeneration is a major cause of chronic low back pain. This represents a leading cause of disability with significant economic and social burdens. The IVD consists of an inner nucleus pulposus (NP) surrounded by the annulus fibrosus (AF) tissue. The third structure that characterizes the IVD is the hyaline articular cartilage, located at the endplates between the IVD and the vertebral bodies.<br /> The gelatinous NP is an avascular tissue containing a highly organized extracellular matrix rich in proteoglycans and collagens with few dispersed cells. In this respect, the NP cells reside within hypoxic conditions, since no vasculature enters the NP. Furthermore, disc cells actively regulate the homeostasis of the extracellular matrix by several cytokines and growth factors acting in an autocrine and paracrine fashion.<br /> Conventional treatments for IVD degeneration are limited, since conservative or surgical therapies do not restore IVD tissue properties. Since the IVD has very limited healing capacity, regenerative medicine (e. g. by injection of cells) may represent a promising therapy for treatment of disc degeneration. As such, IVD repair strategies require an appropriate cell source that is able to regenerate the damaged NP tissue such as progenitor and stem cells.<br /> Progenitor cells do have the advantage over terminally differentiated cells that they maintain their multipotent differentiation and self-renewal potential in-vivo and in-vitro under appropriate conditions. Furthermore, these cells play an important role in the development and homeostasis of the IVD tissue. Recently, progenitor cells that are positive for the angiopoietin-1 receptor (Tie2) were identified in a variety of mammalian’s NP including humans. These cells, which express aggrecan and collagen type II, were shown to have progenitor-like multipotency.<br /> Tie2 is also known as CD202b, a cellular membrane receptor tyrosine kinase of the Tie family. This receptor contains immunoglobulin-like loops and an epidermal growth factor (EGF) similar domain 2. Expressed mainly in endothelial cells, the angiopoietin groups of ligands, upon binding to their receptor Tie2, are known to regulate angiogenesis. Tie2 signaling appears to be critical for endothelial smooth muscle communication and vascular maturation.<br /> The contribution of Tie2 to IVD homeostasis, however, is still poorly understood. Here, we isolated primary nucleus pulposus cells (NPC) from human discs after trauma and sorted these for the Tie2 marker. Here the Tie2+ fraction of cells is suggested to represent the nucleus pulposus progenitor cells (NPPC) population. To demonstrate the stemness of the Tie2+ cells, trilineage (adipogenic, chondrogenic and osteogenic) differentiation assays are performed for the Tie2- and Tie2+ cell populations and then their ability to form colonies in methylcellulose-based medium is monitored. A second aim will be to address the reported difficulties to maintain the phenotype of NPPC in culture and to test different cell culture conditions to maintain and eventually expand these cells <em>in-vitro</em> in culture.</p> <h2>Technological progress in the advanced therapy field</h2> <p>The iPSpine consortium will make fundamental progress in testing induced pluripotent stem cells (iPSCs) as an alternative in IVD degeneration treatment strategy, studying them and gathering evidence that they are more appropriate in the hostile environment of the degenerative IVD than currently employed mesenchymal stem cells (MSCs). Smart instructive biomaterials will be developed that can be employed during manufacturing of the iPSC-based therapeutic product. They also serve as the ideal carrier to deliver the iPSC-based therapy to the patient and provide mechanical support during the regenerative process. iPSpine aims to fill several gaps in the development of a cell-based therapy for LBP identified by the World Health Organization. More in details: which type of cells is to be used, the mechanisms of action, which patients will benefit most (personalized medicine approach), what is the optimal timing to apply the stem cells and what is the best way to deliver and track the cells. The consortium will have access to advanced bioreactor technology, which will better mimic the diseased environment of the tissue of interest. As such, an improved and more thorough screening of possible strategies will be possible, whereby the consortium can improve the strategy to have a better chance of success when ready for clinical implementation.</p> <h2>Acknowledgements of funding sources</h2> <p>The iPSpine project has received funding from the European Union’s Horizon 2020 research and innovation program iPSpine under the grant agreement # 825925 (www.ipspine.eu). The silk work mentioned has been financed by the Gebert Rüf Foundation project «Silk-o-Disc» Project # GRS-X028/13 and the Swiss National Science Foundation Project # 310030E_192674 / 1. The work mentioned in conjunction with the ACL under mechanical stretching has been financed by a start-up grant of the CABMM platform of the University of Zurich.</p></p>
<p class="article-footer">
<a class="literatur" data-toggle="collapse" href="#collapseLiteratur" aria-expanded="false" aria-controls="collapseLiteratur" >Literatur</a>
<div class="collapse" id="collapseLiteratur">
<p><strong>1</strong> GBD 2017 Disease and Injury Incidence and Prevalence Collaborators: Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet 2018; 392(10159): 1789-1858 <strong>2</strong> Wieser S et al.: Cost of low back pain in Switzerland in 2005. Eur J Health Econ 2011; 12(5): 455-67 <strong>3</strong> Markatos K et al.: The anatomy of the ACL and its importance in ACL reconstruction. Eur J Orthop Surg Traumatol 2019; 23(7): 747-52 <strong>4</strong> Frauchiger DA et al.: Genipin-enhanced fibrin hydrogel and novel silk for intervertebral disc repair in a loaded bovine organ culture model. J Funct Biomater 2018; 9(3). pii: E40 <strong>5</strong> Frauchiger DA et al.: Differentiation of MSC and annulus fibrosus cells on genetically engineered silk fleece-membrane-composites enriched for GDF-6 or TGF-β3. J Orthop Res 2018; 36(5): 1324-33 <strong>6</strong> Gantenbein B et al.: Developing bioreactors to host joint-derived tissues that require mechanical stimulation. Encyclopedia of Tissue Engineering and Regenerative Medicine 2019; doi: 10.1016/B978-0-12- 801238-3.65611-8</p>
</div>
</p>
Das könnte Sie auch interessieren:
Frühe Unterwassertherapie nach totaler Hüft- und Knieendoprothese
Eine frühzeitige Durchführung der Unterwassertherapie mit wasserdichtem Folienverband ab dem vierten postoperativen Tag ist im Allgemeinen sicher, wirksam und ohne Nebenwirkungen.
Aktuelles zur anteromedialen Knieinstabilität
Kombinierte Verletzungen des medialen Kollateral- (MCL) und des vorderen Kreuzbandes (VKB) zählen zu den häufigsten multiligamentären Knieverletzungen. Bei vermeintlich isolierten VKB- ...
Arthroskopische Behandlung von Glenoidrandfrakturen
In der Literatur werden die Begriffe knöcherne Bankart-Läsion und Glenoidrandfraktur häufig synonym verwendet, obwohl sie sich hinsichtlich Pathomechanismus und Therapie deutlich ...