<p class="article-intro">Legg-Calvé-Perthes is a hip disorder occurring in children, which causes deformity of the femoral head. For the surgical treatment of severe cases, a so-called femoral head reduction osteotomy (FHRO) can be performed in which a wedge needs to be resected to restore the sphericity of the joint. Although very good outcomes after FHRO have been reported, the surgery is rarely performed because preoperative planning and surgical execution are very challenging. Due to advances in computer-assisted surgery, new treatment concepts for FHRO could be developed which enable safer and more precise execution.</p>
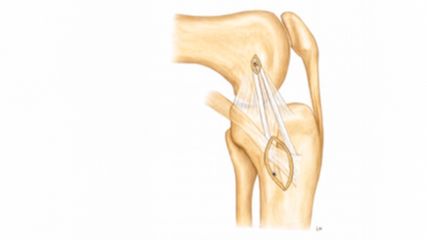
<p class="article-content"><div id="keypoints"> <h2>Keypoints</h2> <ul> <li>Computer-assisted surgery can even drive the development of completely new treatment concepts such as for LCP.</li> <li>The computer-assisted treatment approach comprises a 3D preoperative planning stage in which the surgery is simulated in a patient-specific fashion.</li> <li>Based on the simulated surgery, patient-specific navigation instruments can be designed and 3D-printed.</li> <li>The instruments permit a more precise and safer execution of the surgery. Therewith it may help to further improve the results and hopefully encourages a more prevalent future employment of the FHRO worldwide.</li> </ul> </div> <p>Legg-Calvé-Perthes (LCP) is a relatively common orthopaedic disease in paediatric hip disorders, with a prevalence of 1 case per 1200 children.<sup>1</sup> It has an onset between 5–10 years of age and it is caused by a disturbance of perfusion of the femoral head, leading to avascular necrosis (death of bone tissue) and deformations of the femoral head.<sup>2</sup> Revascularisation is slow and the necrotic bone undergoes fatigue fracturing, leading to flattening of the contour to an ellipsoid shape with an extra-large horizontal and a small vertical diameter. The femoral deformity initiates a reactive adaptation of the acetabulum.<br /> Traditional treatment concepts for LCP range from conservative methods trying to keep the deformation as small as possible with mechanical unloading the joint,<sup>3</sup> to surgical methods trying to improve containment and therewith enlarge the load distribution area.<sup>4</sup><br /> Possible conservative treatments are adductor tenotomy, bracing and physical therapy. Traditional surgical methods, in turn, are femoral varus osteotomy, which realigns the femoral head relative to the hip socket, as well as pelvic osteotomies, to achieve adequate coverage of the femoral head. In one-third of the cases, however, the deformity of the head and the reactive acetabular deformity are too large and exceed the possibilities of traditional treatment concepts.<br /> Deeper understanding of blood supply in the femoral head encouraged the development of a more effective surgical technique,<sup>5</sup> which permits to directly correct the femoral head morphology, rather than indirectly compensating overall joint functionality by altering the femoral shaft or pelvis. The so-called femoral head reduction osteotomy (FHRO) allows reshaping the head to its anatomical spherical shape by resection of the necrotic or deformed central part (Fig. 1).<sup>6</sup> The determination of the optimal correction, however, including definition of the segment to be removed and direction of the cuts to achieve the resection, describes an exceedingly difficult 3D (three dimensional) geometrical problem.<br /> Conservative preoperative planning, which is based on analysis of plain radiographs or single computed tomography (CT) scans, cannot sufficiently capture the 3D problem. Moreover, it lacks appropriate tools for precise surgical implementation of the planned correction. These may be reasons why the number of cases treated with FHRO remains worldwide low considering the prevalence of LCP.<sup>4</sup><br /> Our previous work on computer-assisted surgery methods for complex orthopaedic interventions in upper<sup>7-11</sup> and lower<sup>12</sup> extremities paved the ground for the development of a new computer-assisted FHRO treatment method. We would like to describe the computer-assisted FHRO treatment approach on the basis of an example case of a LCP patient with severe deformation on the left femoral head. The patient was a 15-year-old female pupil. The severe deformity of her femoral head caused pain and forced her to give up rhythmic gymnastics, which she did semiprofessionally since the age of 6 (Fig. 2A, B).<br /> The treatment involves two components, computer-assisted preoperative planning in which every step of the FHRO is computer-simulated in 3D, and intraoperative navigation through patient-specific instruments (PSI) permitting a precise realisation of the planned correction.</p> <h2>Preoperative planning</h2> <p>In a first step, a 3D reconstruction of the pathological bone is obtained by segmenting CT data (Fig. 2B) using a commercial software (Mimics, Version 19; Materialise, Leuven, Belgium). The resulting 3D model (Fig. 2C) can then be imported into our in-house developed preoperative planning software CASPA (Computer Assisted Surgical Planning Application, Version 5; Balgrist CARD AG, Zurich, Switzerland), which is capable of precisely simulating each step of the surgical procedure in 3D. Thereby, the optimal osteotomy parameters can be established in a quantitative and objective fashion.<br /> As shown in Fig. 3, the planning procedure is composed of the following steps: the placement of the osteotomy planes (Fig. 3A) such that the wedge (red) can be resected and the lateral fragment (purple) can be mobilized (Fig. 3B). The reduction of the mobile fragment to achieve a spherical femoral head is shown in Fig. 3C. In this example case, we used a mirror-model of the healthy contralateral femur as a correction goal (Fig. 3D). In case of bilateral deformities, we employ so-called statistical shape models (SSMs) to predict the pre-morbid anatomy. A SSM is built from femora models from a representative set of individuals in order to learn the anatomical variation between different subjects. This summation of all possible shapes can then be fitted to a new patient for predicting the healthy shape of the femoral head without pathological deformation. Restoring the sphericity of the femoral head is the main objective in the preoperative planning. Another challenge is to define the osteotomy planes without compromising the blood supply of the femoral head.<sup>5</sup> Additionally, a decent remaining neck diameter on the femoral shaft, approximately 1/3 of the pathological neck diameter, needs to be considered as well to ensure overall stability (Fig. 3C).</p> <h2>Patient-specific navigation instruments and surgical execution</h2> <p>The CASPA software additionally permits the design of PSI which guarantee a very precise execution of surgical tasks such as drilling, cutting, and reduction (Fig. 4). Each PSI is represented as 3D model and has a dedicated position and function in the preoperative planning. For instance, the PSI for guiding the osteotomies of the FHRO are located anteriorly on the proximal femur shaft. They integrate a cutting slit in which the sawblade can be entered to perform the osteotomy exactly as planned. They key idea of PSI is the undersurface of the instrument base, which guarantees to place the instruments in the surgery on the bone exactly on the same position as preoperatively planned. This can be achieved by computer algorithms which are capable of moulding the undersurface as a negative of the bone surface. Afterwards, a selective laser-sintering device (3D printer) is used to manufacture the PSI as CE-conform medical products (manufacturer: Medacta SA, Castel San Pietro, Switzerland). A biocompatible polyamide PA2200 is used as the raw material for printing.<br /> Before surgery, the PSI are verified on a true-size replica of the pathological femur (see Fig. 4B) and sterilised with conventional steam pressure sterilisation.<br /> The FHRO surgery starts with an exposure of the hip using the surgical dislocation approach,<sup>13</sup> including a trochanteric osteotomy, which enables full access to the joint. The trochanter and hence muscles and ligaments are reattached after the resection. The first PSI is then applied to the femoral shaft and shifted proximally until the optimal position can be found (Fig. 5). The first osteotomy is performed by inserting the saw blade into the slit of the PSI (lateral cut, Fig. 5A). Afterwards, the second PSI is applied to complete the wedge resection (medial cut, Fig. 5B), followed by the reduction of the mobile fragment to its planned position (Fig. 5D). The fragment is secured with three lag screws to the medial part, while two further screws secure the trochanter fragment (Fig. 6).</p> <h2>Discussion and outlook</h2> <p>The progress in technology, especially in computer simulation and 3D printing, has enabled the application computer-assisted preoperative planning and surgical navigation in hip surgery. Above all, we see the benefits particularly for FHRO: The precise planning of the osteotomies, which are required to reconstruct the sphericity of the head, is hardly possible on conventional, plain radiographs or single CT scans due to the limitations of 2D based methods. To date, the proposed computer-assisted treatment approach has been successfully applied in five LCP cases. Furthermore, we have initiated a comprehensive clinical trial to investigate the effectiveness of the method more systematically and in a larger setting.<br /> Previous studies have already shown that the application of PSI can result in a significantly improved surgical execution compared to the freehand technique.10 Therefore, we strongly expect to observe equivalently high precision in PSI-navigated FHRO. Moreover, the more controlled execution of the osteotomies reduces technical failures, which is particularly advantageous in complex interventions. Furthermore, the application of PSI has shown to reduce surgery time,<sup>10</sup> which is beneficial for the patient and reduces costs.<br /> However, there are also limitations to this method. The preoperative time effort to create the 3D simulation and to design the PSI is - with up to 5 hours on average - still very high. Consequently, costs for PSI design amount up to 3000 EUR and manufacturing costs of 600 EUR have to be considered as well. Unfortunately, insurance companies do not reimburse these expenses, because the tariff structure in the Swiss health care system is not yet prepared to consider costs of innovative, new technology within a reasonable time. Another drawback is the long production lead time of 2 weeks, required for 3D printing of the PSI.<br /> Future work will address the development of computer algorithms to automate the planning process such that planning times and costs can be reduced. Furthermore, the consideration of additional image modalities such as MRI will enable the integration of cartilage models into preoperative planning.<br /> The herein presented new computerbased treatment concept for LCP permits sophisticated 3D preoperative planning and a simpler, safer and more precise surgical execution.</p> <p><img src="/custom/img/files/files_datafiles_data_Zeitungen_2018_Leading Opinions_Ortho_1804_Weblinks_lo_ortho_1804_s6_fig1.jpg" alt="" width="450" /></p> <p><img src="/custom/img/files/files_datafiles_data_Zeitungen_2018_Leading Opinions_Ortho_1804_Weblinks_lo_ortho_1804_s7_fig2-4.jpg" alt="" width="1432" height="2178" /></p> <p><img src="/custom/img/files/files_datafiles_data_Zeitungen_2018_Leading Opinions_Ortho_1804_Weblinks_lo_ortho_1804_s8_fig5.jpg" alt="" width="2165" height="782" /></p> <p><img src="/custom/img/files/files_datafiles_data_Zeitungen_2018_Leading Opinions_Ortho_1804_Weblinks_lo_ortho_1804_s8_fig6.jpg" alt="" width="300" /></p> <p><em>This work was funded by the Promedica Stiftung Switzerland and the Deutsche Arthrosehilfe.</em></p></p>
<p class="article-footer">
<a class="literatur" data-toggle="collapse" href="#collapseLiteratur" aria-expanded="false" aria-controls="collapseLiteratur" >Literatur</a>
<div class="collapse" id="collapseLiteratur">
<p><strong>1</strong> Wada I et al.: [Bone disease with pain. Legg-Calvé-Perthes' disease (LCPD)]. Clin Calcium 2008; 18(29): 239-48 <strong>2</strong> Ziebarth K et al.: Residual Perthes deformity and surgical reduction of the size of the femoral head. Oper Tech Orthop 2013; 23(3): 134-9 <strong>3</strong> Wild A et al.: Die nichtoperative Therapie des Morbus Perthes. Orthopäde 2003; 32(2): 139-45 <strong>4</strong> Siebenrock KA et al.: Head reduction osteotomy with additional containment surgery improves sphericity and containment and reduces pain in Legg- Calvé-Perthes disease. Clin Orthop Relat Res 2015; 473(4): 1274-83 <strong>5</strong> Gautier E et al.: Anatomy of the medial femoral circumflex artery and its surgical implications. J Bone Joint Surg Br 2000; 82(5): 679-83 <strong>6</strong> Ganz R et al.: Extended retinacular soft-tissue flap for intra-articular hip surgery: surgical technique, indications, and results of application. Instr Course Lect 2009; 58: 241-55 <strong>7</strong> Vlachopoulos L et al.: Three-dimensional corrective osteotomies of complex malunited humeral fractures using patient- specific guides. J Shoulder Elbow Surg 2016; 25(12): 2040-7 <strong>8</strong> Vlachopoulos L et al.: Computer-assisted planning and patient-specific guides for the treatment of midshaft clavicle malunions. J Shoulder Elbow Surg 2017; 26(8): 1367-73 <strong>9</strong> Schweizer A et al.: Three-dimensional correction of distal radius intra-articular malunions using patient-specific drill guides. J Hand Surg Am 2013; 38(12): 2339-47 <strong>10</strong> Schweizer A et al.: Computer-assisted 3-dimensional reconstructions of scaphoid fractures and nonunions with and without the use of patient-specific guides: early clinical outcomes and postoperative assessments of reconstruction accuracy. J Hand Surg Am 2016; 41(1): 59-61 <strong>11</strong> Vlachopoulos L et al.: Three-dimensional postoperative accuracy of extra-articular forearm osteotomies using CT-scan based patient-specific surgical guides. BMC Musculoskelet Disord 2015; 16: 336 <strong>12</strong> Wirth S et al.: Computer aided three-dimensional surgical planning with patient-specific instruments for accurate correction of malaligned bones of the foot and ankle. Fuss und Sprunggelenk 2015; 13(2): 123-32 <strong>13</strong> Ganz R et al.: Surgical dislocation of the adult hip a technique with full access to the femoral head and acetabulum without the risk of avascular necrosis. J Bone Joint Surg Br 2001; 83(8): 1119-24</p>
</div>
</p>