Non-cutaneous toxicity of targeted therapy for melanoma
Authors:
Dr. med. Egle Ramelyte
Prof. Dr. med. Reinhard Dummer
Dermatologische Klinik
Universitätsspital Zürich
E-Mail: egle.ramelyte@usz.ch
Sie sind bereits registriert?
Loggen Sie sich mit Ihrem Universimed-Benutzerkonto ein:
Sie sind noch nicht registriert?
Registrieren Sie sich jetzt kostenlos auf universimed.com und erhalten Sie Zugang zu allen Artikeln, bewerten Sie Inhalte und speichern Sie interessante Beiträge in Ihrem persönlichen Bereich
zum späteren Lesen. Ihre Registrierung ist für alle Unversimed-Portale gültig. (inkl. allgemeineplus.at & med-Diplom.at)
Cutaneous melanoma accounts for most of deathsrelated to skin cancer. BRAF and MEK inhibitors are practice-changing drugs with overall response rates of up to 70% and median overall survival reaching 33 months. The majority of patients develops at least one adverse event (AE) during therapy; however, most AEs can be well managed and do not require treatment discontinuation.
Keypoints
-
Up to 70% of metastatic melanoma patients with BRAF mutation respond to targeted therapy with BRAF and MEK inhibitors.
-
Along the cutaneous toxicities, the most common adverse events (AEs) are pyrexia and related toxicities, increased laboratory parameters, hepatotoxicity and gastrointestinal events.
-
Some AEs are class effects and occur within all three approved drug combinations, while others are substance- related and are more common within an individual BRAFi or MEKi.
-
Most AEs can be managed with symptom control, treatment interruption or dose reduction and, in some cases, treatment discontinuation.
Overview of non-cutaneous adverse events
Around 40% of cutaneous melanomas harbor a BRAFV600 mutation, which can be targeted with BRAF and MEK inhibitors (BRAFi and MEKi).1 First introduced to the market in 2011, currently there are three combinations of BRAFi and MEKi: vemurafenib (V) and cobimetinib (C), dabrafenib (D) and trametinib (T), and encorafenib (E) and binimetinib (B).
Along with high response rates to BRAFi and MEKi, nearly every treated patient experiences at least one adverse event (AE),2–4 most commonly dermatological AEs or general disorders, such as fever or fatigue. The incidence of grade 3–4 AEs varies between the combinations and is observed in 69% of V+C,2 58% E+B3 and 46–56% of D+T4 treated patients.
The most common serious AEs [SAEs] with V+C are dermal and epidermal conditions (13,3% of cases), while for D+T and E+B it is general disorders (10,6% and 12%, respectively).5 In a systematic analysis, reported odds ratios (ROR) for SAEs were compared among the three combinations. Dermatological AEs and acute kidney injury were higher with V+C than E+B or D+T; pyrexia and elevated C-reactive protein (CRP) lower with D+T than V+C or E+B, while ROR for gastrointestinal AEs and Guillain-Barré syndrome were higher with E+B than V+C or D+T.5 These differences are related to pharmacologic properties of the substances. E inhibits BRAFV600 kinase with a similar concentration as D, but much lower than V (half-maximal inhibitory concentration [IC50] 0,35nM, 0,65nM and 10nM, respectively). The IC50, at which these drugs inhibited the growth of BRAF-mutated cell lines, was also lower for E (<40nmol/L) than for D (<100nmol/L) or V (<1000nmol/L). The dissociation half-life (t1/2diss), which describes the durability of target inhibition, is longer for E than for D or V (>30h, 2h and 0,5h, respectively).6 For MEK inhibition, T has the lowest MEK1 and MEK2 IC50 values (0,7–0,9nM), compared to C (0,95nM and 199nM) and B (12–46nM).7 These measures influence drug-dosing intervals and thus exposure and off-target effects. Moreover, drugs have different stabilizing additives, which also shape the toxicity profile.
AEs related to BRAFi and MEKi treatmentcan be divided into class effects, observed with all three drug combinations, and substance-related AEs, which occur more commonly with a certain drug or combination. Despite the high frequency of AEs, most can be managed symptomatically and/or with dose adjustment. Indeed, AEs led to dose interruption in 46–57%, dose reduction in 12–28% and treatment discontinuation in 12–16% of patients. In phase III trials, dose intensities for the three combinations ranged between 94 and 100% with a treatment duration of 8,8 month for V+C, 10–11 months for D+T and 11,7 months for E+B.8,9
Class effects
The mitogen-activated protein kinase (MAPK) pathway has an important function in the majority of healthy tissues, such as gastrointestinal tract tissues, myocardium, kidney, immune cells or tissues of the eye. Class effects are related to the mechanism of action, e.g. MAPK pathway inhibition, and not to an individual drug. Thus, with different frequencies, they are observed in all combinations of BRAFi and MEKi.
Gastrointestinal events
The most common gastrointestinal (GI) toxicities during BRAFi and MEKi therapy are diarrhea, nausea and vomiting, which can occur with abdominal pain or bleeding. The pathophysiology of GI toxicity is not entirely clear; however, in normal GI mucosa, the MAPK pathway is activated through epidermal growth factor receptor (EGFR) and it negatively regulates chloride excretion. The pathway blockage could lead to increased chloride secretion10 and thus secretory diarrhea. As it is a class effect, diarrhea can occur as early as first day after treatment start.11 E.g. in E+B patients, median time to occurrence of nausea and diarrhea was 29 days (range for nausea 1–614, diarrhea 1–534) and of vomiting 57 days (range 1–607).12
All GI AEs are common with all drug combinations, the most common being diarrhea (18–57%), nausea (20–41%) and vomiting (14–30%).2–4 With E+B, the most common GI AE is nausea (41%), diarrhea (36%), vomiting (30%) and abdominal pain (17%). With D+T, nausea occurred in 35%, vomiting in 29% and diarrhea in 23%, while with V+C the most common GI AE was diarrhea (60%), followed by nausea (41%) and vomiting (24%). Despite being relatively common, grade 3–4 GI AEs occur in 0,3–6,5%, most commonly due to diarrhea (V+C).
Usually managed symptomatically, nausea, diarrhea and vomiting led to dose interruption in 8%, 4% and 7% of cases, respectively, while in up to 1% of patients, diarrhea may also lead to treatment discontinuation.12 GI AEs usually resolve within days to weeks; however, some, such as diarrhea, recur in up to 40% of cases. Along symptomatic GI AEs, asymptomatic hepatic AEs that manifest as liver function test (LFT) elevation are also very frequent. In D+T, an increase in alanine aminotransferase (ALT) is observed in 14%, in aspartate aminotransferase (AST) in 12% and in gamma-glutamyl transferase (GGT) in 11% of the patients. These enzymes increase in 26%, 24% and respectively 22% of V+C,2 and 11%, 8% and 15% of E+B3 treated patients. However, besides asymptomatic LFT elevation, severe drug-induced liver injury was reported for BRAFi and MEKi.13 Pancreatic enzyme elevation is less common, however, D+T led to increase in alkaline phosphatase (AP) in 7,4% and was not reported for lipase, V+C in 17% for AP and in 3,6% for lipase, and E+B in 8,3% for AP and 2,1% for lipase.
Inflammatory and rheumatic events
The MAPK signaling pathway and MEK inhibitors have effects in stimulating the immune response and shaping inflammation.14,15 Thus not surprisingly, inflammatory AEs, such as arthralgia, myalgia and other rheumatic AEs, affect up to a third of treated patients. These AEs can develop early after treatment initiation, while with E+B they occur at a median time of 85 days after treatment start (range 1–708). Arthralgia occurs in 38,1% of V+C patients,2 26,6% of D+T patients,4 and 25,5% of E+B patients.3 However, grade 3–4 cases are uncommon and occur in 0,5–2,4% of cases with all combinations. Myalgia affects 14–19% of BRAFi and MEKi treated patients. As with arthralgia, most cases are grade 1–2; however, cases of rhabdomyolysis have been reported.16 In the phase III clinical trials, none of the rheumatologic events led to discontinuation, but in 2% of cases a dose interruption or adjustment was needed.12
Ocular events
Most, if not all ocular events are class events of MEKi. Their pathogenesis is not entirely clear; however, the suggested etiology is a dysfunction of the metabolism and fluid regulation or the retinal pigment epithelium.17 As the MAPK signaling pathway regulates the density of aquaporins between retinal pigment epithelial cells, MEK inhibition may change the permeability and lead to fluid accumulation in the retina and a serous neuroretinal detachment. As it is a class effect, it is very common.
In COLUMBUS trial, serous retinopathy was observed in 20% of E+B patients in a median time of 38 (1–532) days after treatment initiation. It was asymptomatic (grade 1) in 12%, grade 2 in 5% and grade 3 in 3% of patients.12 With V+C, ocular AEs occurred in 27% of patients, more than half of which (57%) were asymptomatic and diagnosed during regular examination.11
A prospective observational study assessed ocular AEs in patients receiving E+B. All patients experienced serous neuroretinal detachment and edema. However, in this prospective trial, more than half of the patients (69%) reported symptoms, mostly visual disturbances.18 Interestingly, with continuation of treatment, the retinal volume and thickness decreased to levels below baseline without functional loss.
Serous neuroretinal detachment is the most common ocular AE, however, other AEs, such as ocular inflammatory AEs like uveitis and conjunctivitis, have also been reported. Ocular AEs did not lead to discontinuation in E+B patients and only rarely to dose interruption or reduction.12 They resolve quickly, however, recurrence is reported in up to 25% of patients.11
Cardiovascular events
The MAPK pathway is important in the protection and recovery of cardiomyocytes.19 Interestingly, mice that lack members of the MAPK pathway ERK(extracellular signal regulated kinases)-1/2 have normal heart size and function.However, they develop larger myocardial infarcts after coronary artery ligation.20 Thus, in humans, along with treatment with BRAFi and MEKi, another cardiologic risk factor, such as hypertension or diarrhea-related electrolyte disbalance, may be needed for the cardiotoxicity to manifest. Cardiovascular events occur with both, BRAFi and MEKi, however, BRAFi mostly lead to QT prolongation, while MEKi lead to left ventricular ejection fraction (EF) drop. These AEs usually present within 2–5 months but can manifest as early as the next day after treatment initiation.3, 11, 12 In a combination, QT prolongation was more common with D+T (4,5%) than V+C (2%) and was not reported in any of the E+B treated patients. EF drop was most common in V+C, (11,7%), followed by D+T (8,3%) and E+B (5,7%).3 Grade 3 events, which represent an EF of <40% or a decrease by >20% since baseline, occurred in 1–3,7%.
Among other cardiovascular AEs, hypertension was reported in 29%, 16% and 11% patients receiving D+T, C+V and E+B, respectively. Cardiovascular events seldom lead to treatment discontinuation (1–3%) and resolve in majority of cases. However, recurrence rates are up to 24%.4, 21
Mincu et al. conducted a systematic review and meta-analysis of clinical trials, comparing approved BRAFi and MEKi combinations to BRAFi monotherapy. Five randomized clinical trials including 2317 patients were selected and analyzed. Compared to BRAFi monotherapy, treatment with BRAFi and MEKi combination was associated with increased risk for pulmonary embolism (risk ratio [RR]: 4,36; 95% CI: 1,23–15,44; p = 0,02), LVEF drop (RR: 3,72; 95% CI: 1,74–7,94; p<0,001) and arterial hypertension (RR: 1,49; 95% CI: 1,12–1,97; p=0,005). The RR for QTc prolongation was similar among the treatments.22
Nephrotoxicity
BRAF was shown to be expressed in the glomerular podocytes of mice.23 It interacts directly with phospholipase C epsilon 1 (PLCe1), mutations of which are known to be involved in nephrotic syndrome.24 Dabrafenib-treated podocyte monolayers demonstrated reduced levels of PLCe1 and nephrin expression and increased albumin permeability, which was restored after dabrafenib withdrawal.
Data regarding nephrotoxicity is scarce as AEs are rather uncommon; however, increased serum creatinine, acute kidney injury (AKI) or electrolyte disturbances were reported with all substances. AKI was observed in 1,1%, 2,8% and 1,6% of D+T, V+C and E+B patients. Grade 3–4 nephrotoxicity events were also rare (1,1–1,6%).
Substance effects
Pyrexia and related events
The pathogenesis of pyrexia under treatment with BRAFi and MEKi is still not clear. Defined as core body temperature >38,0°C, it often occurs during the first month of therapy and shows lower incidence and recurrence rates as treatment is continued.11,25 Pyrexia is reported to occur with all treatment combinations. However, while the incidence of fever and fatigue was 18% and 28,6% with E+B and 28,7% and 36,8% with V+C,2 it was much more prevalent with D+T: 55,1% of patients developed pyrexia and 31,4% developed associated events, including fatigue.4
Importantly, approximately a quarter of patients develop fever-associated symptoms, such as chills, without an elevated body core temperature.25 Pyrexia and associated events are mostly grade 1, but grade 3–4 pyrexia is reported in 1,2–4,6% and fatigue in 1,1–4,5% of patients.12 With D+T, pyrexia led to dose interruption in 26% and 66%, and dose reduction in 17% and 44% of patients with grade 1 and 2 events, respectively.25 Pyrexia often shows recurrence with a median of 2 and maximum 15 events with D+T. Indeed, 21% of patients who developed recurrence with D+T had 4 or more fever episodes, 13% had 3 events, while 49% of patients only had one event.25
Neurotoxicity
The MAPK pathway drives Schwann cell dedifferentiation upon normal axonal signaling and is required for repair process following nerve injury.26 AEs of the nervous system are rare with BRAFi and MEKi. However, transient Bell’s palsy, a mononeuritic variant of the Guillain–Barrê syndrome (GBS), was reported in 8% of Encorafenib patients.6 Interestingly, it was less common in the E+B combination,3 which is likely due to decreased paradoxical MAPK pathway activation in a combination therapy.
Conclusion
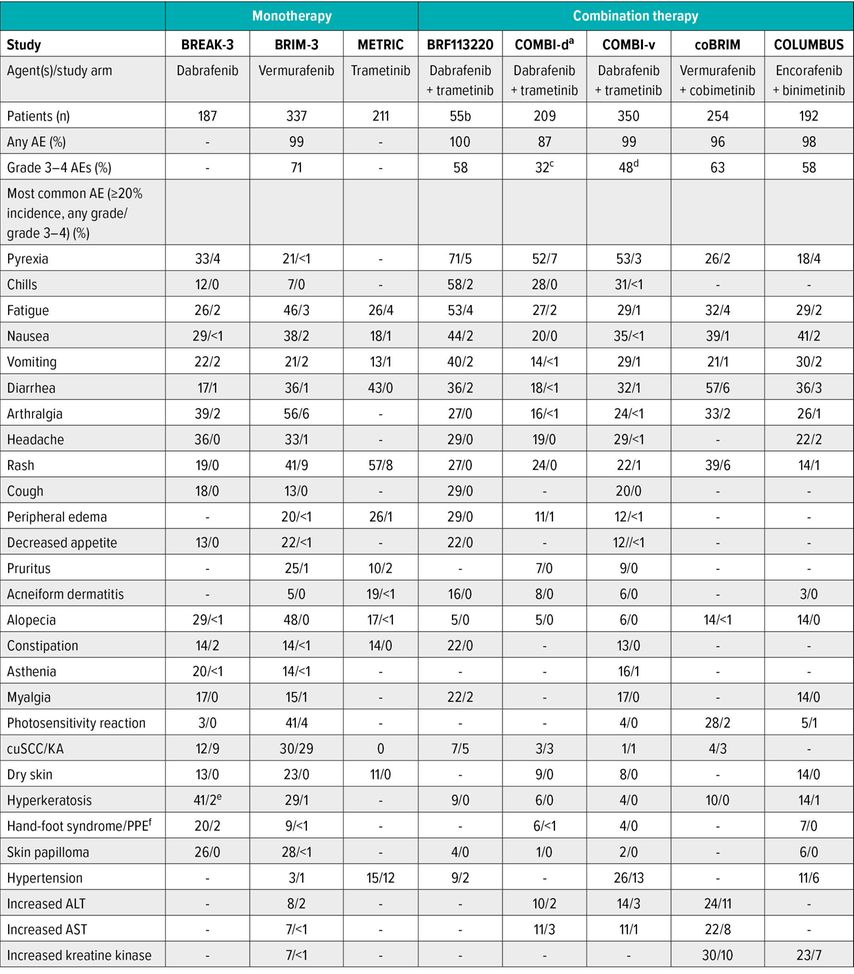
Table 1 gives an overview of the most common AEs associated with BRAFi and MEKi as monotherapy or in combination. Most of the BRAFi and MEKi associated AEs can be managed by symptom control, dose interruption or reduction. While class effects are shared between the combinations, in case of intolerable substance associated AEs, treatment can be changed to a different combination with a more suitable toxicity profile.
Tab. 1: Overview of the most common AEs associated with BRAFi and MEKi as monotherapy or in combination. -, data not reported; AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CuSCC, cutaneous squamous cell carcinoma; KA, keratoacanthoma; PPE, palmarplantar erythrodysesthesia. aData quoted are for treatment-related AEs rather than all AEs. bData for dosage arm of dabrafenib 150 mg twice daily plus trametinib 2 mg once daily; this is the approved dosage. cData are for grade 3 events; one grade 4 event occurred in the dabrafenib and trametinib group (pancytopenia), and three occurred in the dabrafenib and placebo group (thrombocytopenia, febrile neutropenia, hypokalemia). dData are for grade 3 events; three grade 4 events occurred in three patients in the dabrafenib and trametinib combination arm (headache, asthenia, increased AST), and five occurred in five patients in the vemurafenib arm (hypertension, constipation, increased ALT, cuSCC). eHyperkeratosis included acanthoma, acrochordon, actinic keratosis, keratosis pilaris, lichenoid keratosis, papilloma, seborrheic keratosis, and skin papilloma. fHand-foot syndrome includes the terms “palmarplantar erythrodysesthesia”, “plantarpalmar hyperkeratosis” and “palmoplantar keratoderma.” Modified from Daud A, Tsai K27
Literature:
1 Cancer Genome Atlas Network: Cell 2015; 161(7): 1681-96 2 Ascierto PA et al.: Lancet Oncol 2016; 17(9): 1248-60 3 Dummer R et al.: Lancet Oncol 2018; 19(10): 1315-27 4 Robert C et al.: N Engl J Med 2015; 372(1): 30-9 5 Meirson T et al.: Cancers (Basel) 2020; 12(6) 6 Delord JP et al.: Clin Cancer Res 2017; 23(18): 5339-48 7 Cheng Y et al.: Molecules 2017; 22(10) 8 Hamid O et al.: Cancers (Basel) 2019; 11(11) 9 Heinzerling L et al.: ESMO Open 2019; 4(3): e000491 10 Keely SJ, Barrett KE: Am J Physiol Cell Physiol 2003; 284(2): C339-4811 Dreno B et al.: Ann Oncol 2017; 28(5): 1137-44 12 Gogas HJ et al.: Eur J Cancer 2019; 119: 97-106 13 Gravbrot N et al.: Case Rep Oncol Med 2019; 2019: 3051945 14 Verma V et al.: Nat Immunol 2021; 22(1): 53-66 15 Dummer R et al.: Oncoimmunology 2017; 6(8): e1335843 16 Muto Y et al.: Melanoma Res 2018; 28(2): 151-4 17 Jiang Q et al.: Int J Mol Med 2009; 23(6): 771-7 18 Urner-Bloch U et al.: Eur J Cancer 2016; 65: 130-8 19 Lips DJ et al.: Circulation 2004; 109(16): 1938-41 20 Rose BA et al.: Physiol Rev 2010; 90(4): 1507-46 21 Long GV et al.: Lancet 2015; 386(9992): 444-51 22 Mincu RI et al.: JAMA Netw Open 2019; 2(8): e198890 23 Chaib H et al.: Am J Physiol Renal Physiol 2008; 294(1): F93-9 24 Boyer O et al.: J Med Genet 2010; 47(7): 445-52 25 Menzies AM et al.: Ann Oncol.2015; 26(2): 415-21 26 Harrisingh MC et al.: EMBO J 2004; 23(15): 3061-71 27 Daud A, Tsai K: Oncologist 2017; 22(7): 823-33
Das könnte Sie auch interessieren:
Aminosäuren – Booster für die Wundheilung?
Für den Wundheilungsprozess ist je nach Heilungsprozess die richtige Kombination aus Kohlenhydraten, Fetten und Proteinen sowie aus Mineralien, Spurenelementen und Vitaminen essenziell. ...
Ein haariger Fall mit irreversiblen Folgen
Bestimmte Formen von Alopezie scheinen in jüngster Zeit explosionsartig zuzunehmen, wobei die genauen Ursachen bislang noch nicht vollständig geklärt sind. Handelt es sich dabei um eine ...
Die menschliche Haut in der modernen Kunst
Dr. Ralph Ubl, Professor für neuere Kunstgeschichte an der Universität Basel, stellte sich der schwierigen Herausforderung, einem Raum voller erwartungsvoller Dermatologen das Organ Haut ...