Visualizing focal chronic inflammation in multiple sclerosis: paramagnetic rim lesions and slowly expanding lesions
Author:
Alessandro Cagol, MD
Translational Imaging in Neurology (ThINk) Basel, Department of Biomedical Engineering, Faculty of Medicine, University Hospital Basel and University of Basel
Multiple Sclerosis Centre, Departments of Neurology, Clinical Research and Biomedicine, University Hospital and University Basel
Research Center for Clinical Neuroimmunology and Neuroscience Basel (RC2NB), University Hospital Basel and University of Basel
Department of Health Sciences, University of Genova, Italy
E-Mail: alessandro.cagol@unibas.ch
With increasing awareness of the progression independent of clinical relapses, imaging plays an increasingly important role in the monitoring of people with multiple sclerosis. This article gives a short overview of the most important imaging techniques and their limitations in clinical practice.
Keypoints
-
One of the key determinants of multiple sclerosis progression is ongoing chronic inflammation, which can occur across all disease stages.
-
Chronic active lesions can be detected in vivo using MRI as paramagnetic rim lesions and slowly expanding lesions.
-
Paramagnetic rim lesions are highly specific for multiple sclerosis, and provide both diagnostic and prognostic information.
-
Slowly expanding lesions reflect persistent lesion activity over time and help predict disability worsening.
Imaging chronic inflammation
For a long time, the most common form of multiple sclerosis (MS) has been viewed as a disease with two clearly distinct phases. An initial relapsing-remitting phase was considered predominantly inflammatory and clinically driven by relapses, while a later secondary progressive phase was thought to reflect a largely separate neurodegenerative process.
Accumulating evidence now challenges this dichotomous model. The clinical course of MS is better understood as a continuum, shaped by the simultaneous contribution of multiple pathophysiological mechanisms whose relative weight varies across individuals and over time. In this framework, inflammatory and neurodegenerative processes are not sequential, but overlap from the earliest disease stages.1
A key clinical manifestation of this concept is progression independent of clinical relapses (PIRA), which is increasingly recognized as a frequent phenomenon across all MS phenotypes.2 This is particularly evident in the current therapeutic era, in which highly effective disease-modifying therapies successfully suppress acute inflammatory activity, yet disability progression may still occur. To better understand and ultimately address progression, there has been a growing emphasis on moving from a purely clinical definition toward a biologically based characterization of disease mechanisms.1 Among the processes thought to underlie progression, the persistence of compartmentalized, chronic inflammation within established lesions has emerged as a major contributor.3 Advanced imaging techniques now allow this smoldering inflammatory activity to be visualized in vivo, offering insights that extend well beyond conventional lesion counting.
Lesion formation and evolution in MS
MS lesions form following breakdown of the blood-brain barrier, with infiltration of peripheral inflammatory cells and focal demyelination. From this initial stage, lesions may follow different evolutionary paths. Some lesions show partial remyelination, resulting in so-called shadow plaques, while others become inactive and remain relatively stable over time. Importantly, a subset of lesions exhibits persistent inflammatory activity, predominantly localized at the lesion border. Histopathological studies have described these as chronic active (or smoldering or mixed active-inactive lesions), characterized by a hypocellular, demyelinated core surrounded by a rim of activated microglia and macrophages. These lesions expand slowly over time and are thought to contribute disproportionately to neurodegeneration.4 For many years, their identification was restricted to post-mortem studies; this gap is now increasingly being bridged by neuroimaging techniques.
Imaging chronic active lesions in vivo
Chronic active lesions can be investigated in vivo using complementary imaging approaches. Each modality captures a different aspect of the underlying biology, reflecting either persistent inflammatory activity or its slow structural consequences.3 Susceptibility-based magnetic resonance imaging (MRI) sequences enable the visualization of iron accumulation at the lesion edge, where chronic inflammatory activity is concentrated. Using this approach, chronic active lesions appear as paramagnetic rim lesions (PRLs), characterized by a rim of paramagnetic signal surrounding a T2-hyperintense lesion core. In parallel, conventional MRI can capture the dynamic footprint of chronic lesion activity over time: on longitudinal T1- or T2-weighted imaging, these lesions may show gradual, concentric enlargement and are therefore termed slowly expanding lesions (SELs). Beyond MRI, chronic lesion inflammation can also be assessed using 18kDa translocator protein positron emission tomography (TSPO-PET), which targets the translocator protein expressed by activated microglia and macrophages. While TSPO-PET provides a more direct measure of innate immune activation, its clinical applicability is limited by low spatial resolution, limited availability, and radiation exposure.
Among these approaches, PRLs currently represent the most mature and widely validated imaging marker of chronic active lesions, supported by histopathological correlations and increasing methodological standardization.3
Paramagnetic rim lesions
PRLs are the most extensively studied in vivo correlate of chronic active MS lesions. Pathologically, they reflect iron-laden microglia and macrophages persisting at the lesion edge, surrounding an inactive, demyelinated core. This spatial organization mirrors the hallmark features of chronic active lesions described in histopathological studies.
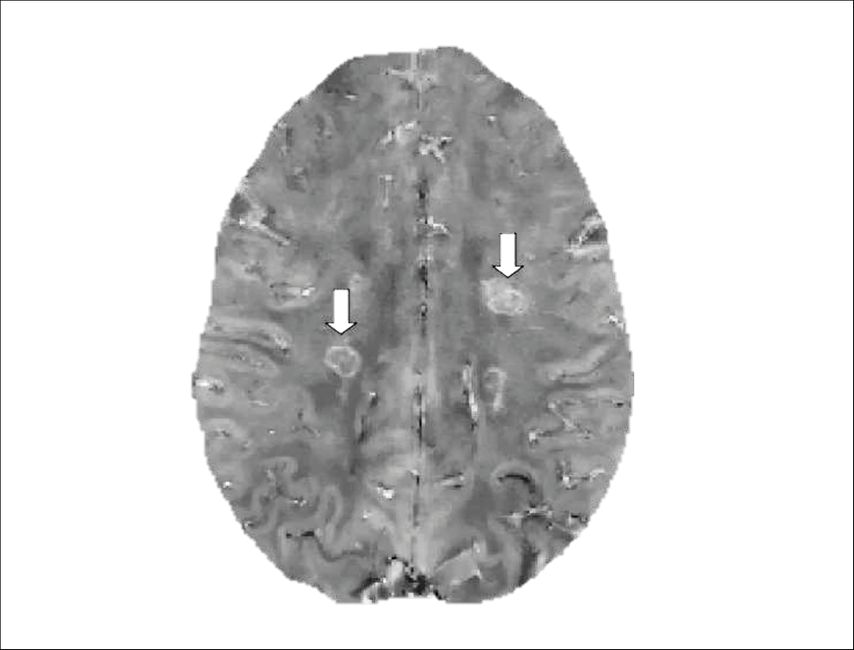
On MRI, PRLs are identified using susceptibility-based sequences, such as susceptibility-weighted imaging (SWI), phase imaging, or quantitative susceptibility mapping (QSM). High-field MRI improves sensitivity, but PRLs can also be detected at 3T and 1.5T with optimized protocols.3
PRLs are observed across all MS phenotypes, including relapsing-remitting, secondary progressive, and primary progressive disease. A recent meta-analysis across published studies reported a prevalence of approximately 51% of patients harboring at least one PRLs, highlighting that these lesions are common rather than exceptional in MS populations.5 Importantly, PRLs are rarely observed in non-MS conditions, a high specificity that has motivated their inclusion in the most recent revisions of the MS McDonald diagnostic criteria as supportive evidence for diagnosis.6
Beyond their diagnostic value, PRLs carry substantial prognostic information. Compared with other lesion types, PRLs are associated with more severe microstructural damage, reflecting more pronounced and persistent tissue injury. At the patient level, their presence correlates with greater neurodegenerative changes, including smaller global and regional brain volumes, as well as higher levels of physical and cognitive disability.3,7 Longitudinal studies further indicate that PRLs are linked to an increased risk of disability worsening, conversion to secondary progressive MS, and PIRA, underscoring their relevance as imaging markers of clinically meaningful disease evolution.3,8
Fig. 1: Example of paramagnetic rim lesions (PRLs) on quantitative susceptibility mapping (QSM). PRLs are indicated by arrows and are characterized by a hyperintense rim reflecting iron accumulation (image modified from Cagol et al. 2023)8
Slowly expanding lesions (SELs)
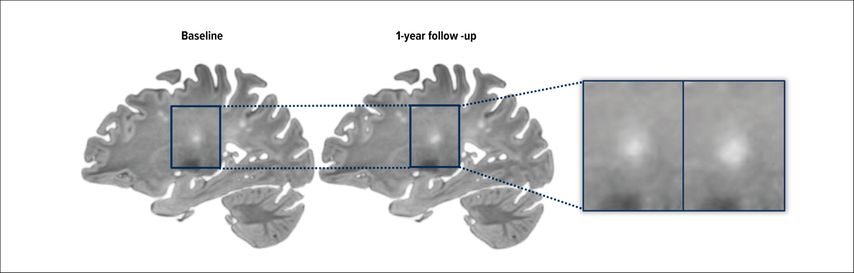
SELs capture the dynamic evolution of chronic active pathology using conventional MRI and are characterized by a steady, concentric expansion of lesion borders over time. Rather than relying on a specific contrast mechanism, SELs are defined by their behavior on longitudinal imaging, where they show slow but persistent radial growth, typically over years. This pattern is considered a structural manifestation of ongoing, compartmentalized chronic inflammation and is thought to contribute directly to disease progression.3
SELs are identified using longitudinal registration of T1-weighted and/or T2-weighted images. Using these approaches, SELs have been reported in the vast majority of patients with progressive MS, with a prevalence of approximately 99%, and in a large proportion of patients with relapsing MS, where they are observed in about 86% of cases.9 SELs are associated with more severe microstructural damage compared with other lesion types. Importantly, the presence and burden of SELs have been shown to predict future disability worsening, supporting their role as imaging markers of clinically relevant, progression-related pathology.10
Treatment effects and the relationship between PRLs and SELs
Despite their growing relevance, evidence on the effectiveness of current disease-modifying therapies in targeting PRLs and SELs remains limited. Available data suggest that these markers of chronic, compartmentalized inflammation are relatively resistant to treatments that primarily suppress acute inflammatory activity.11 More robust and systematic evidence is expected in the coming years as these biomarkers are increasingly incorporated into clinical trials and longitudinal observational studies.
The relationship between PRLs and SELs is also not yet fully understood. While both are considered imaging correlates of chronic active lesions, their overlap is limited. SELs are reported to be approximately two- to eight-fold more frequent than PRLs, and fewer than 20% of SELs spatially co-localize with a paramagnetic rim.12 This limited overlap may reflect several factors, including technical differences, the possibility that PRLs and SELs capture distinct lesion subtypes, or that they represent different stages along a shared lesion developmental trajectory.
Limitations and open challenges
Despite their promise, both PRLs and SELs still face limitations. For PRLs, assessment remains time-consuming and often requires dedicated post-processing and expert visual interpretation. There is still limited standardization of MRI acquisition protocols, sequence parameters, and operational definitions across centers, which can affect reproducibility. In addition, specific training and experience are needed to reliably identify PRLs and distinguish them from artifacts or non-specific iron deposition. For SELs, key challenges include the lack of direct pathological validation and the absence of a universally accepted operational definition. Detection of SELs requires multiple high-quality longitudinal MRI time points and advanced image registration techniques, limiting their applicability in routine clinical practice and in datasets with shorter follow-up or heterogeneous acquisition protocols. Addressing these limitations through methodological harmonization, validation studies, and automation will be essential to fully unlock the clinical potential of these biomarkers.
Looking ahead
Despite several aspects still requiring clarification, it is increasingly expected that PRLs and SELs will substantially change our understanding of MS. By capturing biologically meaningful processes that are only partially reflected by conventional MRI, these markers have the potential to support more accurate prognostic stratification, enable more individualized treatment approaches, and improve patient selection and outcome assessment in clinical trials.
The clinical relevance of this shift is already evident. The recent introduction of PRLs into the updated McDonald diagnostic criteria as supportive evidence for MS diagnosis represents a clear step toward a more biology-driven framework, in which markers of chronic inflammation complement traditional measures of acute disease activity. As evidence continues to accumulate, PRLs and SELs are likely to play an increasingly important role in redefining how disease activity and progression are conceptualized and monitored in MS.
Literature:
1 Kuhlmann T et al.: Multiple sclerosis progression: time for a new mechanism-driven framework. Lancet Neurol 2023; 22(1): 78-88 2 Müller J et al.: Harmonizing definitions for progression independent of relapse activity in multiple sclerosis: a systematic review. JAMA Neurol 2023; 80(11): 1232-45 3 Bagnato F et al.: Imaging chronic active lesions in multiple sclerosis: a consensus statement. Brain 2024; 147(9): 2913-33 4 Dal-Bianco A et al.: Slow expansion of multiple sclerosis iron rim lesions: pathology and 7 T magnetic resonance imaging. Acta Neuropathol 2017; 133(1): 25 5 Martire MS et al.: What is the potential of paramagnetic rim lesions as diagnostic indicators in multiple sclerosis? Expert Rev Neurother 2022; 22(10): 829-37 6 Montalban X et al.: Diagnosis of multiple sclerosis: 2024 revisions of the McDonald criteria. Lancet Neurol 2025; 24(10): 850-65 7 Absinta M et al.: Association of chronic active multiple sclerosis lesions with disability in vivo. JAMA Neurol 2019; 76(12): 1474-83 8 Cagol A et al.: Association of spinal cord atrophy and brain paramagnetic rim lesions with progression independent of relapse activity in people with MS. Neurology 2024; 102(1): e207768 9 Dal-Bianco A et al.: Chronic active lesions in multiple sclerosis: classification, terminology, and clinical significance. Ther Adv Neurol Disord. 2024; 17: 17562864241306684 10 Preziosa P et al.: Slowly expanding lesions predict 9-year multiple sclerosis disease progression. Neurol Neuroimmunol Neuroinflamm 2022; 9(2): e1139 11 Maggi P et al.: B cell depletion therapy does not resolve chronic active multiple sclerosis lesions. EBioMedicine 2023; 94: 104701 12 Elliott C et al.: Lesion-level correspondence and longitudinal properties of paramagnetic rim and slowly expanding lesions in multiple sclerosis. Mult Scler 2023; 29(6): 680-90
Das könnte Sie auch interessieren:
Parkinsonprävention durch Lebensstilmodifikation
Noch bevor die ersten motorischen Symptome der Parkinsonkrankheit auftreten, beginnt die Erkrankung auf biologischer Ebene. Auch wenn bislang keine pharmakologische Neuroprotektion oder ...
Ist die ketogene Diät eine Präzisionsmedizin?
Die ketogenen Ernährungstherapien sind etablierte Behandlungsformen bei Epilepsie. Während sie primär bei therapierefraktären pädiatrischen Epilepsien eingesetzt werden, finden sie ...
Neues aus der Alzheimer’s Disease Drug Development Pipeline
Mit der weltweiten Zulassung der Amyloidantikörper Lecanemab und Donanemab ist erstmals eine kausale Behandlung der Alzheimerkrankheit möglich geworden. Die Behandlung setzt an der ...